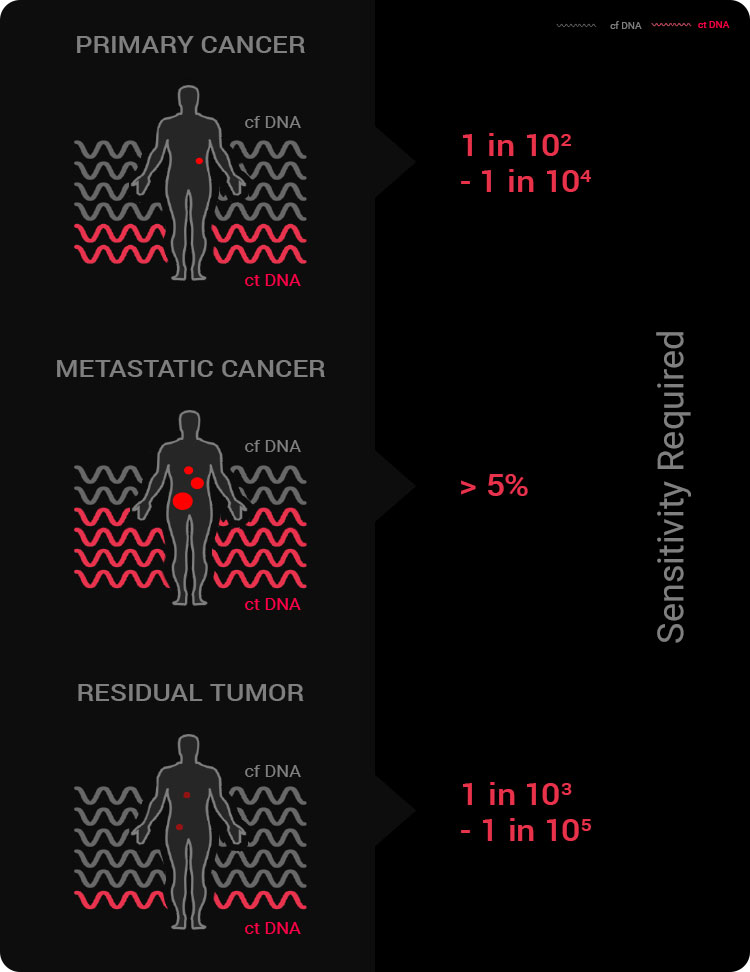
Why is detection sensitivity key to liquid biopsy?
Liquid biopsy: An emerging technology and its limitation.
Data-based precision medicine could be possible if we could accurately analyze molecular information from cancer tissues. The rapid rise of liquid biopsy in cancer diagnosis is the direct result of its non-invasive and economical nature when compared with tissue biopsy. Even more significantly, repetitive tests can be performed and it becomes possible to analyze cancer information. It is especially the circulating tumor DNA (ctDNA-based) liquid biopsy that stands in the spotlight of the liquid biopsy market. ctDNA is more efficient than other biomarkers such as circulating tumor cells (CTCs) or extracellular vesicles (EVs).
While liquid biopsy is rapidly emerging as an alternative to tissue biopsy, it is not yet the gold standard for cancer diagnosis. This is due to the technology limitations of liquid biopsy that prevent a precise analysis of trace amounts of biomarkers from body fluids. If technology that can accurately detect ctDNA in the blood becomes available, ctDNA liquid biopsy will be usable throughout the entire process of cancer diagnosis and eventually could even replace tissue biopsy.
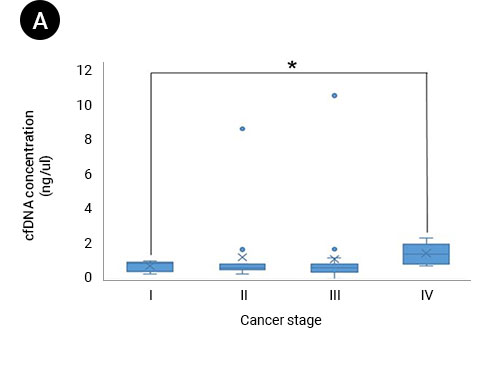
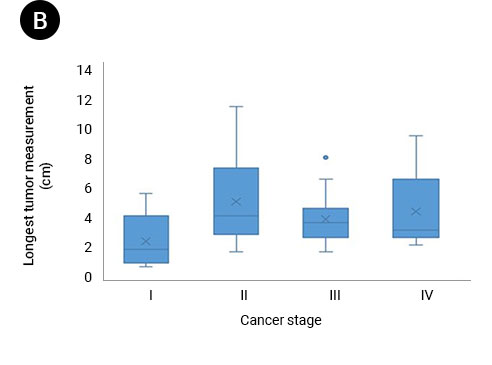

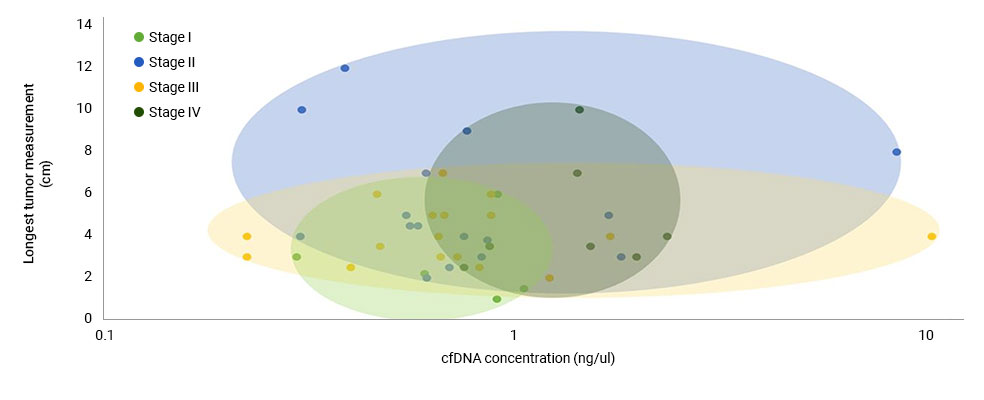
(A) Box and whisker plots of patient ctDNA concentrations compared with cancer stages. The ctDNA concentration in patients with stage I cancer was significantly lower than that in patients with stage IV cancer (P = 0.0149). (B) Box and whisker plots of each patient’s LTM (cm) compared with cancer size. (C) Colored ovals represent the range of ctDNA concentration compared with LTM for each patient based on cancer stage.
Source: Ying-Chi Yang et al, Circulating tumor DNA detectable in early- and late-stage colorectal cancer patients, Bioscience Reports, 2018. July, p. 5.
ctDNA could be used starting at diagnosis in the early stages of cancer development to monitoring in later stages, since it appears at the very beginning of cancer development.
Required detection sensitivity for liquid biopsy
According to the report, a detection sensitivity of 0.01% to 1% is required to analyze ctDNA from early-stage patients, but current liquid biopsy technologies fail to achieve a detection sensitivity of 0.01%. Although ctDNA is an effective specimen throughout the entire cycle of cancer, the technology’s limitations make its clinical use difficult. Some of the latest technologies, such as ddPCR, claim to have reached 0.01%, but in reality the testing methods are overly complex and cannot be applied on a large scale due to economic constraints. Therefore, liquid biopsy could replace tissue biopsy only when it achieves a detection sensitivity of 0.01%.
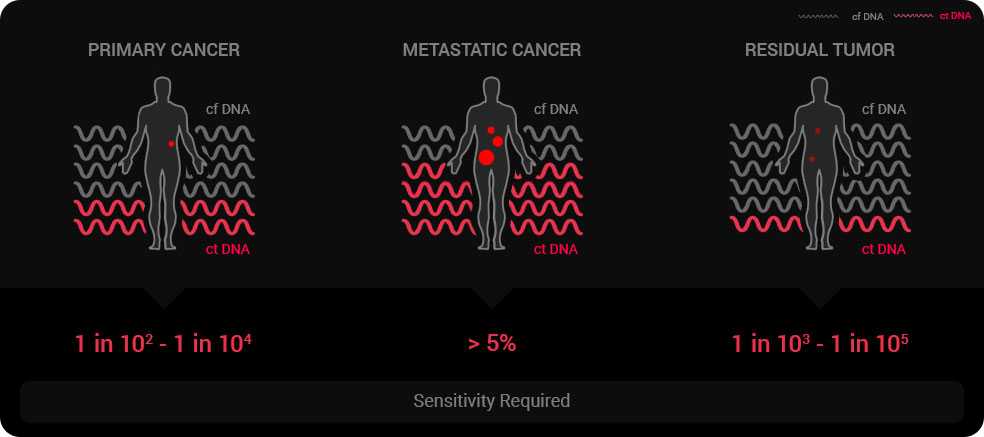
False positive: An obstacle to detection sensitivity improvement
Since detection sensitivity is a limiting factor in enabling the widespread use of liquid biopsy, many companies are competing to improve detection sensitivity. Currently, the practical detection sensitivity of ctDNA that does not sacrifice diagnostic quality remains at around 0.1 to 1%. It is mainly due to false positives that still arise during gene amplification, even when lowering the ctDNA sensitivity threshold.
GENECAST has found a way to overcome this barrier by using ADPS™ which can analyze ctDNA with a sensitivity threshold as low as 0.01%, free of false positives.
ADPS™ overcomes the biggest challenge of liquid biopsy
ADPS™, developed by GENECAST and using a combination of proprietary technologies, has achieved the detection sensitivity of 0.01% necessary for accurate cancer diagnosis using ctDNA. ADPS™ is a qPCR-based technology that is faster and easier to use than other testing methods.
For more information about ADPS™, click “What is ADPS™?” or "How does ADPS™ achieve ultra-high detection sensitivity?"