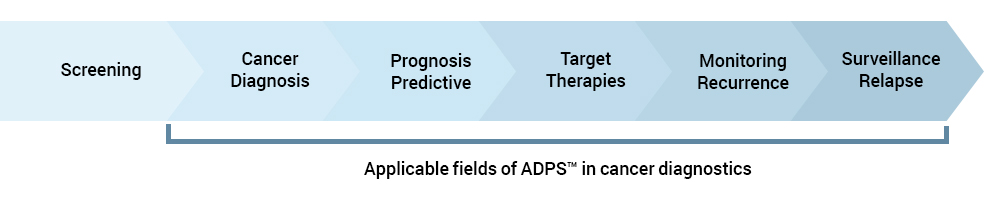
Where can ADPS™ be applied?
ADPS™ covers the entire cancer diagnosis cycle
ADPS™, which can precisely detect ctDNA, can be used during the entire cycle of cancer treatment, from prognosis, diagnosis, and prediction to treatment decision assistance, treatment monitoring, and relapse testing.
With a simple blood test, ADPS™ makes possible what was once only possible via tissue biopsy. Moreover, quantitative analysis becomes possible, facilitating more accurate monitoring and recurrence testing than imaging analysis.
By applying ADPS™, medical organizations can provide the best medical services to patients suffering from various conditions with a single diagnostic method and patients can receive highly efficient and customized medical services of high quality without fear of repeated invasive procedures.
Provides precision medicine for first stage cancer patients
ADPS™, with a detection sensitivity of 0.01%, will lead to more efficient cancer management by significantly expanding the scope of companion diagnostics and precision medicine with liquid biopsy.
Even in patients with a mutation allele ratio of only 0.01%, a quantitative analysis of the cancer gene can help determine the patient’s condition more accurately and make effective treatment decisions regarding drug response and resistance.

Improves the success rate in developing targeted anti-cancer drugs
According to the ARK Investment report, combining co-diagnosis from the initial stage of development of targeted anti-cancer drugs will cut the costs of clinical trials by more than 60% and double the chances of a successful outcome. It means that the success rate of new targeted anti-cancer drugs is closely related to valuable companion diagnostics.
The superior detection sensitivity of ADPS™ can identify more clinical cohorts who have good drug responses, which can reduce costs and time of drug development.
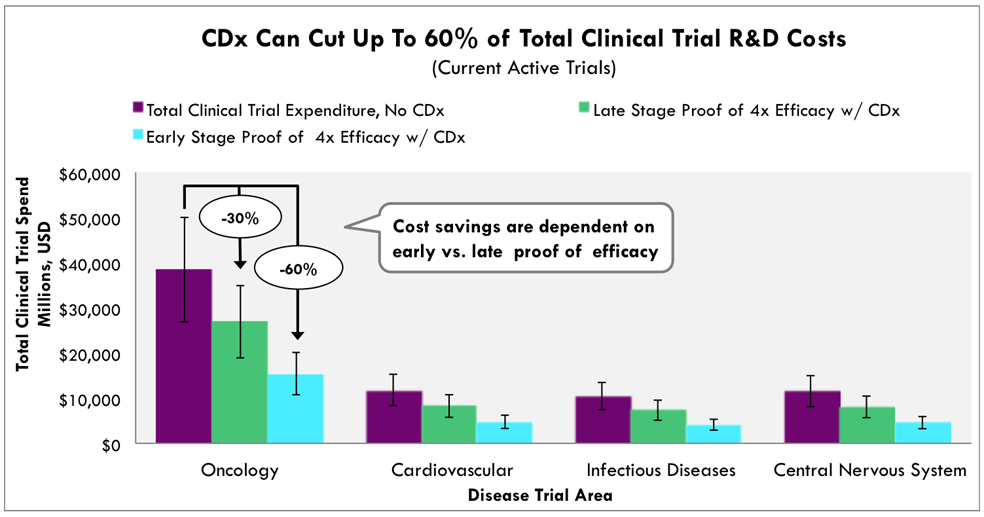
Source: ARK Investment Management LLC
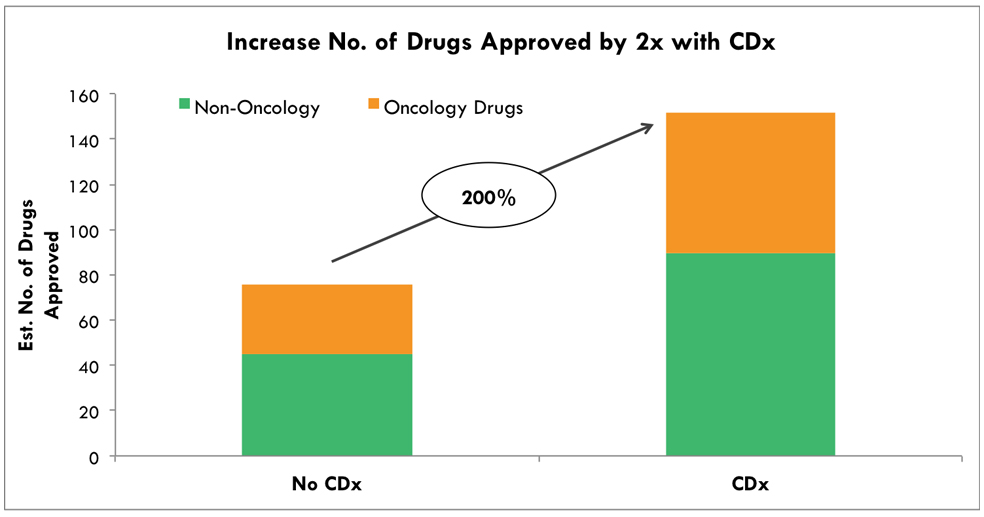
Source: ARK Investment Management LLC
Applicable to various molecular diagnostic fields and diagnostic equipment
ADPS™ a DNA polymerase-based technology which is the core of PCR, can be applied in various molecular diagnostic equipment or fields that require gene discernment.
GENECAST has been working on validating the capabilities of ADPS™ in cancer liquid biopsy, one of the most challenging areas of molecular diagnostics, and has been preparing to enter various fields of molecular diagnostics through licensing and co-development.

Development Plan for Advanced ADPS™ Technology
GENECAST has continually made efforts to expand the scope of ADPS™ and enhance its usability and accuracy. The development of Digital ADPS™ will be completed by 2023. This will be the realization of an early cancer diagnosis system that allows simultaneous screening and diagnosis using liquid biopsy. We are also working to increase the concordance rate between tissue biopsy and liquid biopsy by investigating EVs, mi-RNA, etc.